News and Award
About CMUH
A Physician–Scientist CEO Redefines CAR-T for Solid Tumors
Cancer remains one of the greatest challenges in modern medicine. While advances in surgery, targeted therapy and immuno-oncology have improved outcomes for many patients, those with advanced or treatment-resistant solid tumors often reach a point where therapeutic options become limited. As a neurosurgeon deeply engaged in scientific research, I have come to believe that meaningful medical innovation must do more than introduce new technologies—it must tangibly change what patients experience in their daily lives.
Throughout my career, I have been driven not only by clinical practice, but by a commitment to understanding disease at a deeper biological level. Repeatedly, I encountered patients for whom surgery and standard therapies were no longer enough. That reality compelled me to step beyond the operating room and into drug development, with the goal of helping more patients than any single intervention ever could.
Today, as advanced therapies rapidly enter clinical practice, the central question is no longer whether we can innovate, but whether innovation can truly reach patients in time.
 Dr. Cho, MD, PhD, a neurosurgeon and the hospital president of China Medical University Hospital, has a long-standing commitment to translational research and innovative cell therapy development. (Courtesy of CMUH)
Dr. Cho, MD, PhD, a neurosurgeon and the hospital president of China Medical University Hospital, has a long-standing commitment to translational research and innovative cell therapy development. (Courtesy of CMUH)
The Promise—and Limits—of CAR-T in Solid Tumors
Over the past decade, CAR-T cell therapy has transformed outcomes in hematologic malignancies, offering durable responses in diseases once considered incurable. Yet it is equally important to acknowledge a fundamental limitation in modern oncology: Despite its success in blood cancers, conventional CAR-T therapy has struggled to deliver consistent benefit in solid tumors, which account for more than 90 percent of cancer cases worldwide.
This challenge is especially evident in glioblastoma multiforme (GBM), one of the most aggressive and lethal brain tumors. GBM exemplifies the biological and clinical barriers that limit cell therapy in solid tumors—poor immune infiltration, profound immunosuppression and rapid progression. For clinicians, these realities highlight a deeper systemic problem: therapies that work in theory often fail to reach patients in practice.
Conventional CAR-T approaches typically require lymphodepletion chemotherapy to promote cell expansion, increasing infection risk and excluding many elderly or heavily pretreated patients. Patient-specific manufacturing and MHC-dependent tumor recognition further complicate logistics and delay treatment. For aggressive solid tumors, these constraints significantly limit real-world applicability.
Designing CAR-T Around Solid-Tumor Biology
To overcome these barriers, my team and I asked a simple but disruptive question: What if cell therapy was designed around the biology of solid tumors, rather than adapted from hematologic models?
This question led to the development of CAR001, which through close collaboration with Ever Supreme BioTechnology, has become a leading cell-therapy company in Taiwan that originated as a spin-off from China Medical University Hospital. Our shared objective was not innovation for its own sake, but a therapy grounded in clinical feasibility, safety and unmet patient need.
CAR001 is built on a γδ T-cell platform, enabling tumor recognition in an MHC-independent manner and broadening applicability across diverse patient populations. This γδ T-cell backbone provides innate-like antitumor activity well suited to the heterogeneity of solid tumors.
The therapy incorporates a dual-mechanism design. First, γδ T cells are engineered with a nanobody-based CAR targeting HLA-G, an immune checkpoint molecule frequently overexpressed in advanced solid tumors and associated with immune evasion. By directly recognizing HLA-G, CAR001 enables tumor targeting while bypassing classical antigen presentation pathways.
Second, upon tumor engagement, CAR001 locally secretes a nanobody-based anti–PD-L1 bispecific T-cell engager (BiTE) within the tumor microenvironment. This BiTE simultaneously binds PD-L1 on tumor cells and CD3 on endogenous T cells, recruiting and activating bystander immune cells directly at the tumor site.
Together, this integrated strategy delivers both direct cytotoxic activity and amplification of local antitumor immunity, while minimizing systemic immune activation. From a clinician’s perspective, it represents an effort to reprogram the tumor microenvironment from an immunosuppressive barrier into an immune-active niche—an essential requirement for effective cell therapy in solid tumors.
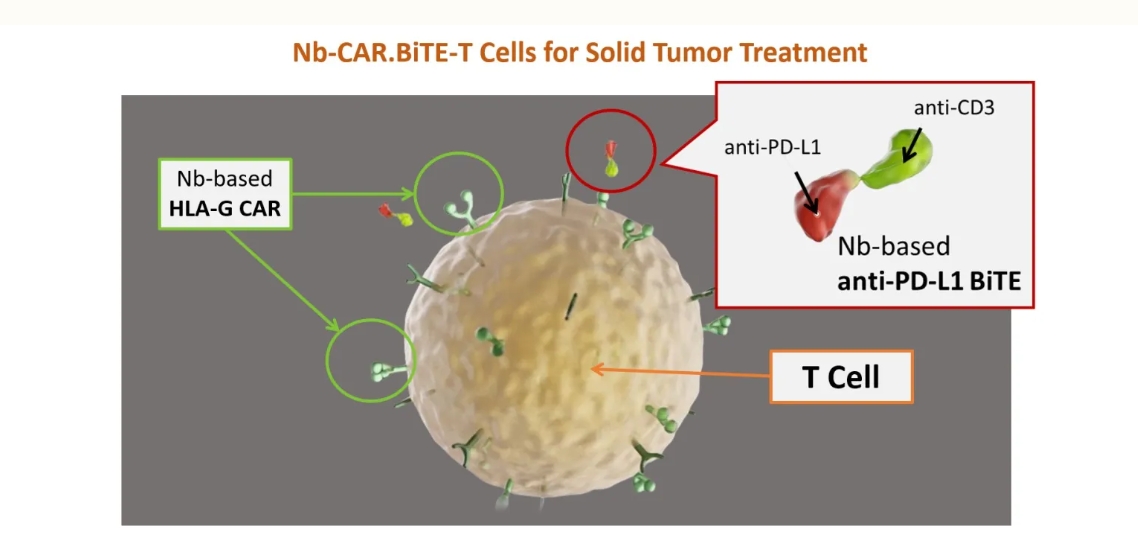
CAR001 is a dual-mechanism γδ CAR-T therapy that directly targets HLA-G–expressing tumor cells while locally recruiting endogenous T cells through PD-L1–directed BiTE activity, thereby overcoming immunosuppression in solid tumors. (Courtesy of CMUH)
Clinical Observations From the Neurosurgical Frontline
As a neurosurgeon, I observed firsthand how these design principles translated into clinical experience.
In an ongoing Phase I/IIa clinical study evaluating CAR001 in patients with refractory or relapsed solid tumors, including GBM, one patient treated at the Cohort 4 dose level demonstrated a 86.02 percent reduction in tumor size on 6 months follow-up MRI imaging. In the context of heavily pretreated GBM, such radiographic tumor shrinkage is uncommon and clinically noteworthy.
Equally important were the functional improvements observed in this patient. At baseline, she presented with markedly slowed speech and impaired expressive ability. Following treatment, her speech gradually improved, approaching near-normal pace and clarity. She regained independent communication and was ultimately able to return to work. For neurosurgeons, these functional outcomes often matter as much as imaging results.
Exploratory cerebrospinal fluid analyses provided additional insight. Approximately three months after treatment, circulating tumor cells in the CSF decreased from 91 cells per 8 cc to 27 cells—an approximate 70 percent reduction. While these findings represent early clinical observations rather than definitive conclusions, the concordance among imaging, functional recovery and biological markers underscores the potential of solid-tumor-specific CAR-T design.
Collaboration as an Engine of Translation
CAR001 reflects the strength of hospital–industry collaboration. Ever Supreme BioTechnology has played a central role in translating clinical insight into scalable manufacturing, regulatory-aligned development and quality systems. This collaborative model has been essential in moving CAR001 from concept to clinical reality and represents a growing strength within Taiwan’s cell-therapy ecosystem.
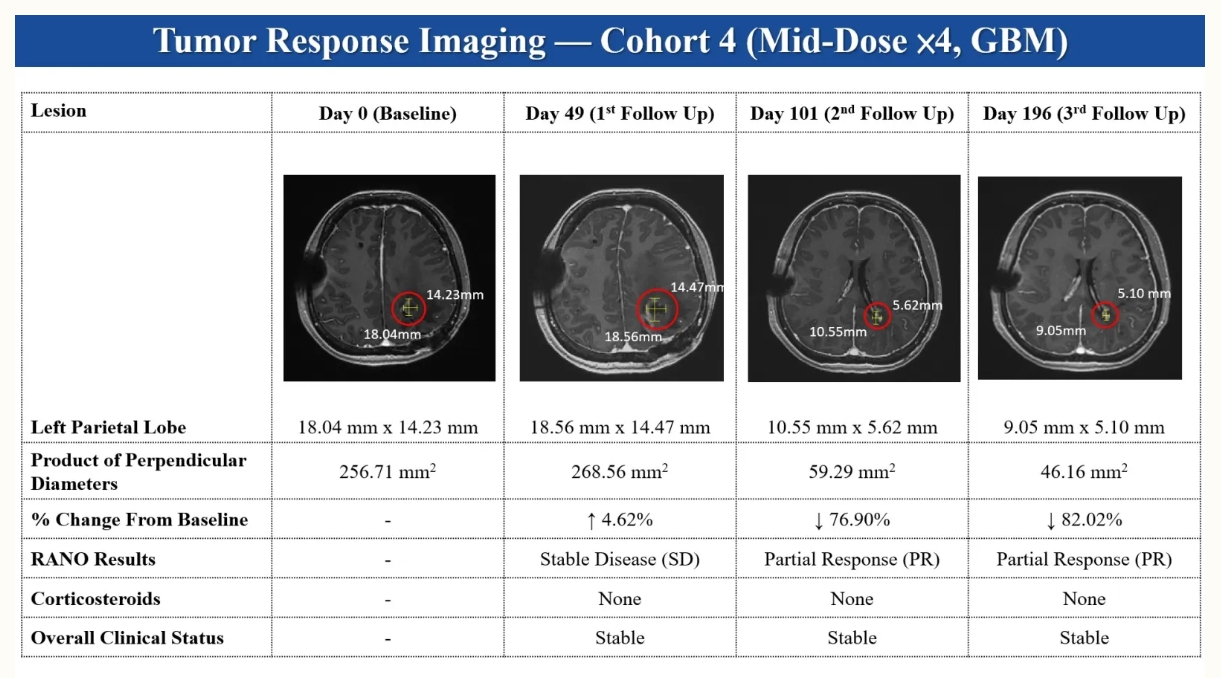 Representative longitudinal MRI images from a patient with recurrent glioblastoma multiforme (GBM) treated with CAR001 in an ongoing Phase I/IIa clinical study. Imaging is shown for illustrative purposes as a physician-observed clinical example and does not represent a definitive efficacy conclusion. (Courtesy of CMUH)
Representative longitudinal MRI images from a patient with recurrent glioblastoma multiforme (GBM) treated with CAR001 in an ongoing Phase I/IIa clinical study. Imaging is shown for illustrative purposes as a physician-observed clinical example and does not represent a definitive efficacy conclusion. (Courtesy of CMUH)
Looking Ahead: Exosome-Based In Vivo EXO 001 CAR-T
While CAR001 represents an important milestone, I believe the next frontier lies in exosome-based in vivo EXO 001 CAR-T—where immune cells are programmed directly inside the patient, much like a vaccine.
This platform, now licensed and transferred to Ever Supreme BioTechnology for regulatory development and future commercialization, eliminates the need for ex vivo cell manipulation. If successfully translated, in vivo CAR-T could reduce complexity, lower cost and expand access to advanced immunotherapy—shifting cell therapy from a bespoke, hospital-intensive procedure toward a standardized, scalable intervention.
A Responsibility Beyond Innovation
My journey from neurosurgery to drug development has been guided by a single motivation: to help more patients than any one operation ever could. CAR001 reflects that commitment—to science, to patients and to the belief that innovation must ultimately serve human life.
The future of cell therapy will not be defined solely by scientific novelty, but by whether advanced treatments can be delivered safely, efficiently and equitably across healthcare systems. For physician–scientists, that challenge is not merely technical—it is an ethical responsibility.
By rethinking CAR-T for solid tumors and building a pathway toward in vivo cell therapy, we are working toward a future where advanced immunotherapy is not only powerful, but practical, scalable and humane.

Hospital–industry collaboration plays a key role in advancing clinically grounded cell therapy innovation within Taiwan’s growing biomedical ecosystem.
Related news from Newsweek: https://www.newsweek.com/a-physician-scientist-ceo-redefines-car-t-for-solid-tumors-11574512



